Rybelsus, Semaglutide
- I. Introduction
- II. Composition of Rybelsus, Semaglutide
- III. How Rybelsus, Semaglutide Works
- IV. Approved Uses of Rybelsus, Semaglutide
- V. Off-Label Uses of Rybelsus, Semaglutide
- VI. Dosage and Administration of Rybelsus, Semaglutide
- VII. Storage and Handling Precautions
- VIII. Interactions with Rybelsus, Semaglutide
- Side Effects and Management
- Warnings and Contraindications
- XI. Careful Administration of Rybelsus, Semaglutide
- XII. Important Precautions with Rybelsus, Semaglutide
I. Introduction
Rybelsus is a medication that includes semaglutide an active ingredient. Semaglutide is in the form of a white to almost white hygroscopic powder. It functions as a glucagon-like peptide 1 (GLP 1) receptor agonist, similar to GLP 1, which is naturally produced in the gut. This action leads to an increase in insulin released by the pancreas in response to food intake.
A. Background of Rybelsus, Semaglutide
To optimize blood sugar (glucose) management in adults diagnosed with type 2 diabetes, healthcare professionals prescribe Rybelsus alongside dietary modifications and regular exercise.
B. Therapeutic Class and Role in Medicine
Type 2 diabetes mellitus can be effectively managed with Semaglutide, an incretin mimetics therapeutic class member.
II. Composition of Rybelsus, Semaglutide
A. Active Ingredient: Semaglutide
Semaglutide is the active ingredient in Rybelsus.
B. Inactive Ingredients and Their Role
Rybelsus contains several inactive ingredients: magnesium stearate, microcrystalline cellulose, povidone, and salcaprozate sodium (SNAC). Magnesium stearate serves as a lubricant during the tablet coating process. Microcrystalline cellulose functions as a binder in the tablet formulation. Povidone acts as a disintegrant. They are aiding the tablets' dissolution in the stomach. Lastly, salcaprozate sodium (SNAC) is an absorption enhancer for semaglutide.
C. Formulation and Availability
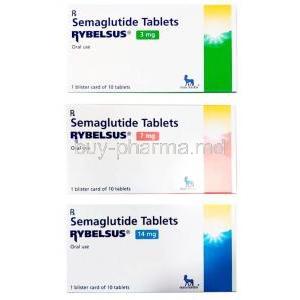
Rybelsus is offered in tablet format with three varying potencies: 3 mg, 7 mg, or 14 mg of semaglutide in each tablet. It is exclusively obtainable through a prescription provided by your healthcare provider.
III. How Rybelsus, Semaglutide Works
A. Mechanism of Action
Rybelsus is composed of semaglutide, serving as its active component. Semaglutide is classified as a glucagon-like peptide 1 (GLP 1) receptor agonist1. Its mechanism of action involves simulating the effects of GLP 1, an endogenous hormone that prompts insulin release and reduces glucagon secretion from the liver2, by activating the GLP 1 receptor. Semaglutide instructs your body to increase insulin production and hinder sugar release. And decelerate digestion2.
B. Impact on Blood Sugar Levels
For adults living with type 2 diabetes seeking improved glycemic control, Rybelsus offers a complementary approach alongside dietary adjustments and physical activity. Including semaglutide allows for increased insulin secretion while suppressing glucagon release. Importantly, these effects operate according to glucose concentrations within an individual's system.
C. Other Potential Therapeutic Impacts
Semaglutide has been demonstrated to possess additional potential therapeutic benefits, including weight loss and cardiovascular risk reduction.
IV. Approved Uses of Rybelsus, Semaglutide
A. Role in Managing Type 2 Diabetes
Rybelsus is prescribed a healthy diet and regular exercise to enhance blood sugar levels in adults diagnosed with type 2 diabetes3. However, it is essential to note that this medication is unsuitable for individuals with type 1 diabetes or for managing diabetic ketoacidosis.
Here are the references
1. FDA approves first oral GLP-1 treatment for type 2 diabetes
2. RYBELSUS® (semaglutide) tablets 7 mg or 14 mg
B. Weight Management: An Additional Benefit
For adults struggling with obesity or excess weight and affected by at least one weight-related condition such as high blood pressure or type 2 diabetes4. The FDA has officially approved semaglutide to address their concerns about managing their weight effectively.
1. FDA approves first oral GLP-1 treatment for type 2 diabetes
2. Medications Containing Semaglutide Marketed for Type 2 Diabetes or Weight Loss
3. FDA Approves New Drug Treatment for Chronic Weight Management
C. Addressing Cardiovascular Risk Factors
Semaglutide has been shown to reduce the risk of major adverse cardiovascular events (MACE) such as heart attack, stroke, or cardiovascular death in adults with type 2 diabetes who have established cardiovascular disease or multiple cardiovascular risk factors.
References:
1. Cardiovascular Safety and Benefits of Semaglutide in Patients With Type 2 Diabetes
3. Semaglutide Approved to Reduce Risk for Major Adverse Cardiovascular Events
V. Off-Label Uses of Rybelsus, Semaglutide
A. Use in Type 1 Diabetes Management
Rybelsus is not suitable for individuals with type 1 diabetes. According to its approval status. Nevertheless. Several studies indicate that semaglutide may hold promise for improving the management of type 1 diabetes.
References:
1. Semaglutide in Type 1 Diabetes: A Review of the Evidence
2. FDA approves first oral GLP-1 treatment for type 2 diabetes
B. Role in Polycystic Ovary Syndrome (PCOS)
Polycystic ovary syndrome (PCOS), a hormonal disorder prevalent among women in their reproductive years, poses significant challenges. It is worth noting that Rybelsus does not have official approval for addressing PCOS; nonetheless, emerging research suggests that semaglutide might hold promise as a management option for affected individuals.
References:
C. Possible Use in Non-Alcoholic Fatty Liver Disease (NAFLD)
Nonalcoholic fatty liver disease (NAFLD) is characterized by the buildup of fat in the liver. Although Rybelsus is not currently authorized to treat NAFLD, certain studies suggest that semaglutide may offer potential advantages in managing this condition.
References:
1. Oral Semaglutide (Rybelsus) for the Treatment of Type 2 Diabetes and Beyond
VI. Dosage and Administration of Rybelsus, Semaglutide
A. Standard Dosage for Adults
The recommended dose for Rybelsus is 7 mg, to be taken once daily for 30 days. After which, it should be increased to 14 mg per day. This medication is intended to be taken orally and can either be consumed with or without food at any time during the day.
B. Adjusting Dosage for Individual Needs
Individual dosage requirements and tolerability may necessitate adjustments to the prescribed amount of Rybelsus. It is important to note that the highest recommended dose of this medication is 14 mg, taken once daily.
C. Instructions for Use
Please swallow the tablets whole and refrain from crushing or chewing them. If a missed dose occurs, kindly take it as soon as possible on the same day. However, if it is already the next day. It is advisable to skip the missed dose and continue with the next scheduled dose at the usual time.
VII. Storage and Handling Precautions
A. Recommended Storage Conditions
It is recommended to store Rybelsus in its original container at room temperature, which should be kept between 68°F to 77°F (20°C to 25°C).
B. Handling Procedures for Safe Usage
To maintain their potency. It is advisable to store Rybelsus tablets in their original blister pack until they are ready for consumption. It is crucial to shield these tablets from both moisture and light.
C. Disposal Guidelines
It is advised to dispose of any unused or expired Rybelsus tablets in accordance with local regulations as it ensures proper management and adherence to guidelines.
VIII. Interactions with Rybelsus, Semaglutide
A. Drug-Drug Interactions
Some drugs can interact with Rybelsus and impact how well it works or increase the chances of experiencing side effects. These drugs include insulin secretagogues (such as sulfonylureas) insulin, GLP 1 receptor agonists (such as exenatide and liraglutide), DPP 4 inhibitors (such as sitagliptin), oral contraceptives, warfarin, CYP inducers (like rifampin) and CYP inhibitors (such as ketoconazole). It is essential to be aware of these potential interactions when taking Rybelsus to ensure its effectiveness and minimize the risk of side effects.
B. Food and Lifestyle Considerations
While consuming Rybelsus, there aren't any specific guidelines about food choices or lifestyle adjustments. However, it remains essential to uphold a nourishing diet and partake in consistent physical activity to effectively manage type 2 diabetes and other ailments that Rybelsus aids in treating.

C. Impact of Comorbid Conditions
Patients with a previous occurrence of pancreatitis or a family background of medullary thyroid carcinoma should exercise caution when considering using Rybelsus. Moreover, it is not advisable for individuals with type 1 diabetes or those seeking treatment for diabetic ketoacidosis. Additionally, patients with renal impairment may need to undergo dose adjustments.
Side Effects and Management
When using Rybelsus, or Semaglutide, you might encounter several side effects. Some common side effects encompass nausea, vomiting, diarrhea, abdominal pain, and decreased appetite. Although these side effects are typically mild and resolve independently, it is essential to be aware of more severe side effects, like pancreatitis, hypoglycemia, and kidney problems, if you experience any side effects while on Rybelsus. Semaglutide, some guidelines can be followed to manage them effectively. These include taking the medication with food to alleviate nausea and vomiting and ensuring adequate fluid intake to prevent dehydration caused by diarrhea.
Warnings and Contraindications
Special precautions should be taken by specific populations when using Rybelsus, Semaglutide. Elderly patients may require a lower dose due to increased sensitivity to the medication. Pregnant women and nursing mothers should exercise caution as the safety of Rybelsus, Semaglutide during pregnancy and breastfeeding is unknown. The safety and effectiveness of this medication in children has not been established. There are also situations where Rybelsus and Semaglutide should be avoided. If you have a history of pancreatitis or gallbladder problems, it is recommended to steer clear of this medication. Additionally, suppose you have a personal or family history of medullary thyroid carcinoma (MTC) or are allergic to any ingredients in Rybelsus or semaglutide. In that case, it is essential to avoid taking it. Suppose you suspect someone else has taken too much Rybelsus or Semaglutide. Be aware of the signs of an overdose, such as nausea and vomiting. Abdominal pain, diarrhea, and hypoglycemia (low blood sugar). In such cases, prompt action should be taken. Regarding managing type 2 diabetes, Rybelsus (semaglutide) is a widely used medication that effectively controls blood sugar levels. However, it is crucial to administer this medication with care by following the instructions provided by your healthcare provider. This article will discuss the significance of regular monitoring, handling missed doses, and making necessary adjustments in dosage while highlighting the essential role healthcare providers play in ensuring successful treatment outcomes.
XI. Careful Administration of Rybelsus, Semaglutide
A. Importance of Regular Monitoring
When taking Rybelsus, regular monitoring is vital. This medication can potentially lead to fluctuations in your blood sugar levels, necessitating adjustments to your dosage or other medications. To ensure your levels fall within a healthy range, your healthcare provider will likely advise regular blood sugar testing. It may also be necessary to monitor your weight and blood pressure when taking Rybelsus. This medication has the potential to result in weight loss and lower blood pressure, which can be advantageous for specific individuals but may require adjustments to other medications if you encounter any side effects.
B. Handling Missed Doses and Dose Adjustments
If you forget to take a dose of Rybelsus, it is crucial to take it as soon as you remember. Nevertheless, if your next dose is almost due, you should skip the missed one and proceed with your regular dosing schedule. Taking additional medication to compensate for the missed dose is not advisable. Your healthcare provider may also suggest modifying your dosage to your blood sugar levels or other factors. You must adhere meticulously to these instructions and refrain from making any adjustments to your dosage without first consulting with your healthcare provider.
C. Role of Healthcare Provider in Successful Treatment
The pivotal role played by your healthcare provider in facilitating fruitful treatment with Rybelsus should not be overlooked. Under their watchful eye, they will closely check your blood sugar levels and other essential indicators to guarantee the medication's effectiveness without any undesirable repercussions. Furthermore, beyond monitoring these factors diligently, you would be pleasantly surprised to learn about their ability to offer invaluable guidance regarding lifestyle adjustments which can remarkably improve your general well-being and mitigate the risks of type 2 diabetes. This relationship could encompass beneficial dietary suggestions, personalized exercises, and possible referrals for seeking specialized care.

XII. Important Precautions with Rybelsus, Semaglutide
A. Avoidance of Potential Drug Misuse
Adherence to your healthcare provider's directions while using Rybelsus is essential to ensure appropriate and successful outcomes. It is imperative not to distribute this medication to others or employ it for any purposes apart from its designated use in treating type 2 diabetes.
B. Monitoring for Signs of Hypoglycemia
If you are taking Rybelsus, you can experience hypoglycemia (low blood sugar). Especially if you are taking other medications that lower blood sugar levels or have kidney problems, some symptoms of hypoglycemia include sweating, shakiness, confusion, dizziness, or fainting. If you notice any of these symptoms while taking Rybelsus, you must seek immediate medical attention.
C. Considerations for Long-term Use
If you are using Rybelsus for an extended duration, It is crucial to regularly monitor your health and promptly inform your healthcare provider of any alterations or worries. The prolonged usage of this medication may elevate the likelihood of specific side effects, such as pancreatitis or kidney issues.
2 . Rybelsus ( Semaglutide ) for Weight loss
3 . Rybelsus for weight loss: how it works
4 . Rybelsus (semaglutide) research information.
5 . FDA Approves New Drug Treatment for Chronic Weight Management, First Since 2014
6 . Semaglutide and Nutrition : How to Optimize Your Treatment
7 . The Future of Type 2 Diabetes Management: Semaglutide, SGLT2 Inhibitors, and Canagliflozin
Rybelsus, Semaglutide FAQ
- Rybelsus side effects?
- Rybelsus weight loss?
- Rybelsus cost?
- Rybelsus coupon?
- Rybelsus versus Ozempic?
- Rybelsus vs Ozempic?
- Rybelsus for weight loss?
- Rybelsus dosage?
- Rybelsus dosing?
- Rybelsus medication?
- Rybelsus foods to avoid?
- Rybelsus vs metformin?
- Rybelsus reviews?
- Rybelsus and weight loss?
- Rybelsus weight loss in non diabetic patients?
- What is Rybelsus?
- Rybelsus dose for weight loss?
- Rybelsus dosage for weight loss?
- Rybelsus price?
- Rybelsus savings card?
- Rybelsus drug class?
- Rybelsus patient assistance program?
- Rybelsus for diabetes?
- Rybelsus copay card?
- Rybelsus uses?
- Rybelsus diabetes?
- Rybelsus vs Jardiance?
- Can Rybelsus be taken at night?
- Rybelsus vs Wegovy?
- Rybelsus vs Ozempic weight loss?
- Rybelsus alternative?
- Rybelsus cost per month?
- Rybelsus class?
Rybelsus side effects?
It should be noted that Rybelsus can potentially induce side effects such as nausea and vomiting. Other possible adverse effects include diarrhea, belly pain, along with constipation. Furthermore, it may cause upset stomach, gas accumulation, and unintended weight loss.
Rybelsus weight loss?
Rybelsus2 has the potential to cause weight loss as an incidental outcome.
Rybelsus cost?
The price of Rybelsus may differ based on the dosage and the pharmacy. It should be noted that the average retail cost for 30 tablets of 7mg is $1,2002.
Rybelsus coupon?
You have the opportunity to obtain coupons for Rybelsus. To gather more details, you may consider contacting your doctor or visiting the manufacturer's website2.
Rybelsus versus Ozempic?
Both Rybelsus and Ozempic have the active drug semaglutide, and they can both result in similar side effects such as nausea, vomiting, diarrhea, belly pain, constipation, upset stomach, gas, and weight loss. Nevertheless, there are particular distinctions between these two medications. For instance, while Ozempic is administered via injection once a week, Rybelsus is taken orally once a day.
Rybelsus vs Ozempic?
Both Rybelsus and Ozempic contain the active drug semaglutide. They can both potentially result in side effects such as nausea, vomiting, diarrhea, stomach pain, constipation, discomfort, flatulence, and weight loss. Nevertheless, there are distinguishing factors between these medications. Notably, Ozempic is administered via injection weekly, whereas Rybelsus is taken orally once daily.
Rybelsus for weight loss?
Rybelsus2 may potentially result in weight loss as a side effect.
Rybelsus dosage?
The initially suggested starting dose for Rybelsus is 3 mg once daily for 30 days. Subsequently, patients are advised to escalate their intake by increasing the dosage to a daily measurement of 7 mg3.
Rybelsus dosing?
For the commencement of therapy with Rybelsus, the suggested starting dose is typically 3 mg administered once daily for a period lasting up to one month. Following this initiation phase, the dose ought to be escalated to 7 mg taken on a daily basis4.
Rybelsus medication?
Rybelsus is an effective medication that is prescribed to adults who are diagnosed with type 2 diabetes.
Rybelsus foods to avoid?
While taking Rybelsus, there is no need to avoid any specific foods. Nevertheless, adhering to a healthy diet and maintaining regular exercise habits is crucial to regulate your blood sugar levels2 effectively.
Rybelsus vs metformin?
Metformin is an additional medication commonly used to treat type 2 diabetes. Its primary mechanism of action involves reducing glucose production in the liver and enhancing insulin sensitivity in the body. It is worth noting that both metformin and Rybelsus can potentially yield similar side effects, such as nausea, vomiting, diarrhea, belly pain, constipation, upset stomach, and gas. Nonetheless, it is essential to recognize some distinctions between these two drugs. Expressly, metformin necessitates oral administration twice or thrice, whereas Rybelsus only requires once-daily oral intake.
Rybelsus reviews?
According to reports. Individuals have experienced weight loss when using Rybelsus. Specifically, some people shed an average of five pounds (equivalent to 2.3 kilograms) while taking Rybelsus 7mg, and even more excellent results were observed with Rybelsus 14mg, where individuals lost an average of 8.1 pounds (approximately 3.7 kilograms)1.
Rybelsus and weight loss?
It is worth noting that Rybelsus1 may potentially result in weight loss, which can be considered as a possible side effect.
Rybelsus weight loss in non diabetic patients?
Although the FDA does not currently approve Rybelsus for weight loss in nondiabetic patients, it appears to possess advantages over Liraglutide, an FDA-approved weight loss medication.
What is Rybelsus?
Rybelsus is an esteemed medication specially designed for treating type 2 diabetes in adult patients231.
Rybelsus dose for weight loss?
It is suggested that individuals begin with a dosage of 3 mg of Rybelsus once a day for 30 days. After this initial month, it is recommended to increase the dose to 7 mg of Rybelsus once daily3.
Rybelsus dosage for weight loss?
The initial dose of Rybelsus as per the recommendation is 3 mg to be taken once daily for a duration of 30 days, following this period. It is advised to raise the dose to 7 mg. Also taken once daily3.
Rybelsus price?
The price of Rybelsus can fluctuate based on the prescribed dosage and the pharmacy chosen. On average, a 30-tablet supply of 7mg tablets is priced at $12001 in retail.
Rybelsus savings card?
There are savings cards readily accessible for Rybelsus. If you seek further details, you may consult with your doctor or visit the official website of the manufacturer2.
Rybelsus drug class?
Rybelsus falls under the category of drugs known as glucagon-like peptide 1 (GLP 1) receptor agonists23.
Rybelsus patient assistance program?
Patient assistance programs are readily available for Rybelsus. It is advisable to consult your doctor or visit the manufacturer's website for further details2.
Rybelsus for diabetes?
Indeed, Rybelsus is a pharmacological intervention to address type 2 diabetes among adult patients.
Rybelsus copay card?
Savings cards for Rybelsus are readily accessible. You may inquire with your doctor or explore the manufacturer's website.
Rybelsus uses?
Rybelsus is an effective medication prescribed for treating type 2 diabetes in adults.
Rybelsus diabetes?
Rybelsus is a medication prescribed to adult patients to manage type 2 diabetes.
Rybelsus vs Jardiance?
Rybelsus and Jardiance are physician-recommended treatments for individuals grappling with type 2 diabetes to regulate their blood sugar levels. It is crucial to note that these medicines operate differently, necessitating a clear distinction between the two.
Can Rybelsus be taken at night?
One crucial aspect to consider is that Rybelsus allows individuals to take it at their preferred time without being constrained by whether or not they have eaten.
Rybelsus vs Wegovy?
Wegovy and Rybelsus are two different brand names for semaglutide, a medication used to treat certain medical conditions. However, they have different formulations and methods of administration.
Rybelsus vs Ozempic weight loss?
Both Ozempic and Rybelsus have been demonstrated to induce weight reduction in individuals diagnosed with type 2 diabetes.
Rybelsus alternative?
Multiple options are at hand regarding managing type 2 diabetes through medications. An insightful discussion with your healthcare provider becomes imperative to ensure a tailored approach suited for you. They possess the necessary knowledge and expertise to guide you toward selecting the most suitable medication.
Rybelsus cost per month?
The price of Rybelsus may vary depending on the prescribed dosage and the pharmacy you choose. At present, the estimated average cost for a one-month supply of 30 tablets containing 7mg each amounts to $1,200.
Rybelsus class?
Rybelsus falls within a category of medications known as glucagon-like peptide 1 (GLP 1) receptor agonists.