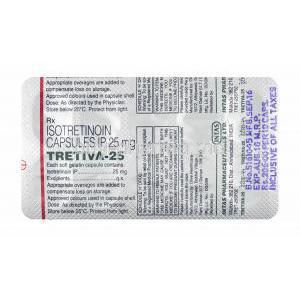
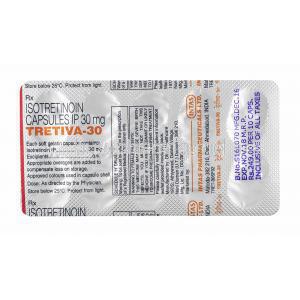
Isotretinoin
Uses
Tretiva is used in the treatment and prevention of acne.
How it Works
Tretiva is known as a form of vitamin A. Tretiva reduces the amount of oil released by oil glands in the skin, and helps the skin renew itself more quickly.
Common Side effects
The USA Food and Drug Administration (FDA) has advised women who are pregnant or planning to become pregnant to NOT consume any form of Tretiva. Tretiva can cause adverse birth defects in babies. Tretiva is known as a category X drug, the FDA has placed a severe advisory against pregnant women or women who are planning to get pregnant against consuming Tretiva. If pregnancy does occur during Tretiva treatment it is advised for the woman to consult with the doctor the option to discontinue the pregnancy as there is a very high risk of birth defects in the baby.
Breastfeeding women are advised against the consumption of Tretiva as this medicine can cause severe health problems in babies.
Some of the potential side effects of Tretiva include the symptoms below;
Bone or joint pain,
Burning,
Eye inflammation,
Nosebleeds,
Redness,
Itching,
Inflammation of the lips,
Skin infection or rash
Expert advice
Rated: 5.0 / 5 based on 5.0 customer reviews.
Posted by Kay on Feb 17, 2020  Verified Purchase
Verified Purchase
Great product BETTER SERVICE
I have ordered many different meds from Robert over 5+ years and they have always been as advertised. In addition to the top rate products Robert's communication regarding any questions have always been very helpful and forthright. Case in point on my most recent order there was some minor difficulty with payment where it took longer to clear on my end than usual. During this time he was very helpful in explaining what was taking so long and how to resolve it
After it finally authorized Robert was very quick to get my order out and it actually arrived sooner than it usually does.
Note: buy-pharma.md does not imply any medical claims from this review.
Posted by Rubin on Jan 8, 2020  Verified Purchase
Verified Purchase
Best Medicine from the Best Company on Earth!
I have been buying various products from Robert at Buy-pharma.md for over 10 years now and I love this company! Whatever you buy from them, you will get it 100% and it will be so fresh and perfect and potent that you will smile when your package comes! I recommend Isotretinoin for anyone who is responsibile (ladies, no pregnancy!!) and wants to CURE acne..period. No creams or time spent on skincare routines, just a pill ALWAYS taken with a high-fat meal does it. Your lips will get dry, but it can de dealt with and it's the only side effect you'll get. The nice thing about the dry lips, is it lets you know you've achieved the levels in your system you need and it's a sign that you've got a potent batch inside you stopping sebum production in its tracks. But with confidence from Buy-pharma.md!
Note: buy-pharma.md does not imply any medical claims from this review.
Posted by Jools on Jan 5, 2020  Verified Purchase
Verified Purchase
Just as effective
These are just as effective as the ones from my doctor. I take ultra-low dose isotretinoin for seborrheic dermatitis. Delivery was quick (about 10-11 days) and discreet. Robert is doing such amazing work in providing Americans access to resonably priced pharma from other countries. Thanks for helping us escape the grip of US pharmacutical corporations. Will keep buying from here.
Note: buy-pharma.md does not imply any medical claims from this review.
Posted by Michalczuk on Oct 30, 2019  Verified Purchase
Verified Purchase
Great product
Great product, works as well as non generic versions. If you suffer with acne I recommend this medicine
Note: buy-pharma.md does not imply any medical claims from this review.
Posted by chavez on May 10, 2019  Verified Purchase
Verified Purchase
Great isotretinoin!!
This medication works great for that stubborn cystic acne. Love it !!
Note: buy-pharma.md does not imply any medical claims from this review.
Posted by Ortega on Apr 25, 2019  Verified Purchase
Verified Purchase
Juvederm Isotretinoin Doxycycline Botox
Awesome pricing for excellent medication
Been using these guys for years and have never been disappointed
Note: buy-pharma.md does not imply any medical claims from this review.
Posted by chavez on Apr 9, 2019  Verified Purchase
Verified Purchase
Great isotretinoin!!
Works great for that nodular cystic stubborn acne. Happy with my results.
Note: buy-pharma.md does not imply any medical claims from this review.
Posted by Ford on Dec 27, 2018  Verified Purchase
Verified Purchase
Isotretinoin
Medication received in about 10 days in pharmaceutical packaging from the manufacturer. It was exactly what I ordered, and definitely professional, with a high standard for the production, just as it would be in a US pharmaceutical company.
Note: buy-pharma.md does not imply any medical claims from this review.
Posted by sandobare on Sep 7, 2018  Verified Purchase
Verified Purchase
tretiva isotretioning
always the best
Note: buy-pharma.md does not imply any medical claims from this review.
Posted by Ford on Aug 25, 2018  Verified Purchase
Verified Purchase
Fast. No problems with product or delivery.
See above.
Note: buy-pharma.md does not imply any medical claims from this review.
Posted by LadyProfessor on Jul 10, 2018  Verified Purchase
Verified Purchase
Acne: Solved
I purchased this when I got fed up with adult acne. At almost 50, I still had daily breakouts of large cysts on my jawline, sebaceous filaments all over my face that oxidized and looked like blackheads, and general scarring associated with the acne. I figured that after suffering from this for 40 years on my chest, face, back, and arms, it was time to do something. I ordered the lowest dose (10 mg) and started it daily. Within 14 days, my skin was transformed. No longer could I scrape waxy sebum from my face and the cysts have not come back in 1 month. For the first time in my adult life, I am free of acne. As a pharmacologist, I know that tretinoin can literally change the protein transcription of my skin genes that cause acne. This is profound. That it begins changing these genes in one week is astonishing, but it is true. Over 16 weeks, sebaceous glands are actually reduced in size, blocking a great deal of the facial "grease" I have put up with for decades. I plan to stay on this indefinitely. For people who think that any drug purchased beyond the US is counterfeit, think again. We actually source most of our active starter material from outside US borders. Also, many of the drugs you get on this site are made in US factories and exported out of the US, so all you are doing is reimporting something we made. Don't fall for selfish scare tactics. Get the drugs you deserve on the terms most favorable to you. I have purchased from this vendor for about 10 years, and I have NEVER ever been let down. Take it from someone who has the ability to actually make drugs...this is legit and worth your time.
Note: buy-pharma.md does not imply any medical claims from this review.